|
Some proteins require other proteins called chaperones in order to correctly fold into their normal 3-D shape.Ĭells use chaperones both to accomplish the original folding of some proteins and to restore the structure of incorrectly folded ones. Proteins have a narrow range of conditions in which they fold properly outside that range, proteins tend to renature. 
If the 3-D structure of a protein depends only on its primary structure and the surrounding environmental conditions, then when the protein isĭenatured and subsequently returned to its native conditions it will spontaneously refold back into its native structure. When normal environmental conditions are reestablished after protein denaturation, almost all proteins can spontaneously refold back into Proteins can denature when the pH, temperature, or ionic concentration of the surrounding solution changes.ĭenatured proteins are usually biologically active. If a protein's environment is altered, the protein may change its shape or even unfold completely, a process called dissociation. A slightly positive hydrogen atom on one water molecule can attract the slightly negative oxygen atom on a different water molecule, leading to the formation of a hydrogen bond. Of the 4 most common elements found in living organisms, nitrogen has the highest electronegativity and, therefore, the greatest affinity for electrons. Although C and H differ slightly in electronegativity, this small difference is negligible, and C-H bonds are considered polar. Polar molecules are electrically neutral but the distribution of charge within the molecule is not uniform. A covalent bond is formed when two atoms share one or more pairs of electrons. A single molecule of oxygen (O2) is held together by two double nonpolar covalent bonds. In a nonpolar covalent bond there is an equal sharing of electrons between two atoms while polar covalent bonds involve an unequal sharing of electrons. 
In general, electronegativity increases from left to right across each row of the periodic table and also increases down each column. Atoms differ in their affinity for neutrons, a property called electronegativity. Because oxygen has a greater electronegativity than hydrogen, water molecules are polar with two partial negative charges near the oxygen atom and one partial positive charge near each hydrogen atom. A water molecule is held together by two single polar covalent bonds. Each energy level can hold a maximum of 2 electrons. In living organisms, chemical energy is stored in high-energy electrons that are transferred from one atom to another in reactions involving oxidation and reduction. Energy levels, which are usually drawn as circles around the nucleus, indicate an electron's energy, while orbitals, which have a variety of three dimensional shapes, indicate an electron's most likely location. Such electrons are said to occupy the same energy level. Because the amount of energy an electron possesses is related to its distance from the nucleus, electrons that are the same distance from the nucleus have the same energy, even if they occupy different orbitals. Because electrons are attracted to the positively charged nucleus, the closer an electron is to the nucleus, the more potential energy it has. Still others, farther away from the nucleus, have other shapes. Some orbitals near the nucleus are spherical (s orbitals), while others are dumbbell-shaped (p orbitals). The second energy level consists of 4 electron orbitals. 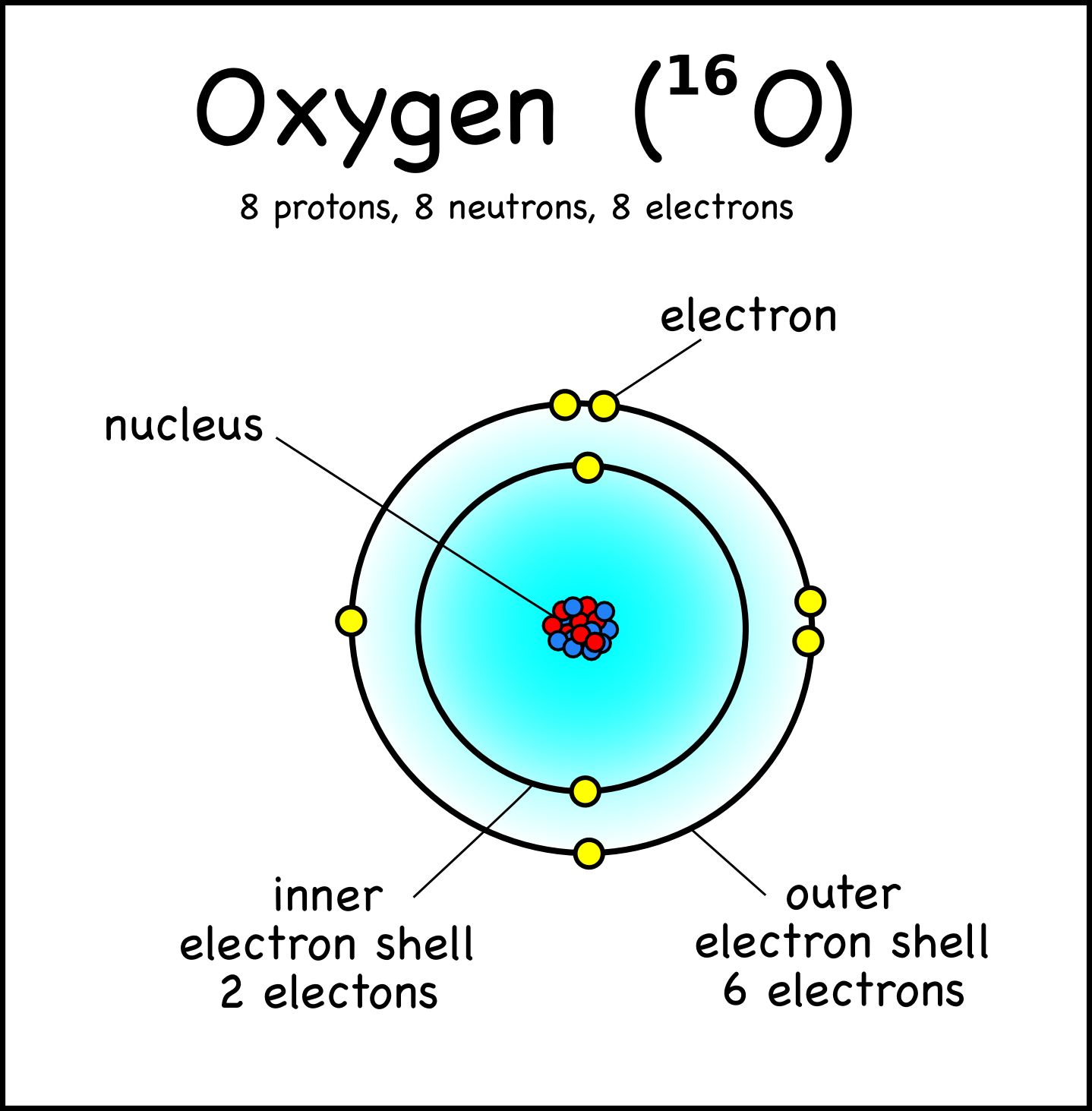
Each energy level consists of 2 or more orbitals. The atom shown in the diagram above has a total of 5 electron orbitals. The atom shown in the diagram has 4 energy levels or shells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
